FALL 2019, THE EVIDENCE FORUM, WHITE PAPER
 Ying Xiao, MHSA Research Associate Evidence Synthesis, Modeling & Communication Evidera |  Ray Gani, PhD Senior Research Scientist and Senior Director Evidence Synthesis, Modeling & Communication Evidera |  Krystal Chen, PhD Consultant Market Access Consulting Evidera |  Can Chen, MSDirector of Access and HEOR Happy Life Technology (HLT) |  Thitima Kongnakorn, PhD Senior Research Scientist Evidence Synthesis, Modeling & Communication Evidera |
Universal healthcare now covers 95% of the population in China and, given limited budgets, healthcare payers are struggling to fund both best medical practice and new innovative medicines. To manage their budgets more effectively, substantial changes have been introduced to the market access process that pharmaceutical companies must follow to achieve reimbursement. Recent critical changes include the introduction of price negotiation and the use of health economics to evaluate the value of new medicines.
These changes present many new challenges for pharmaceutical manufacturers, including the availability of local data, the novel use of health economics, and procedural uncertainties in a fast-changing landscape.
e to download printable PDF
Background
Pharmaceutical companies launching in China aim to have their medicines added to the National Reimbursement Drug List (NRDL). Once on the list, the national basic medical insurance (BMI) covers 50% to 70% of the cost of the medicines. Prior to 2015, this process was haphazard and inconsistent. There were many barriers and uncertainties, which included non-transparent, decision-making processes and long waiting times before listing. Many delays were due to frequent changes in the processes and policy, leaving a significant gap in evaluating new treatments and patchy access with local variation.
The national health action plan, “Health China 2030” launched in October 2016, initiated the trend of moving towards evidence-based health policy making in China. The drivers for the trend were identified as1:
- Greater life expectancy
- An aging population due to prior demographic policies (e.g., one-child rule) in China, which has resulted in relatively few working-age adults able to provide the elderly care
- Rising incidence of non-communicable disease resulting from rapid urbanization and economic growth2
- Expanding healthcare coverage and increasing out-of-pocket payments
These pressures meant that significant reforms and evidence-based policy making were needed to ensure that the provision of healthcare was allocated fairly, consistently, and effectively. This led to the national pilot of price negotiations mainly for innovative but expensive medicines in 2017, transforming the pricing and reimbursement processes in China.
NRDL Updates
The past few years witnessed two major milestones, in 2017 and 2019, in NRDL updates as part of healthcare reform.
2017 NRLD Update: Introduction of Negotiation for Inclusion
In 2017, after a long eight years, the NRDL was updated to add 339 drugs, with a focus on drugs treating catastrophic diseases such as cancer, hematological disorders, and HIV. Products for rare diseases also received more attention than in the past.3,4
The total number of covered drugs increased to 2,535, which represents an increase of 15.4% compared to 2009 when the NRDL was last updated. A breakdown of the therapeutic areas of newly listed drugs is shown in Figure 1.3
Figure 1. Breakdown of Therapeutic Area of Newly Listed Drugs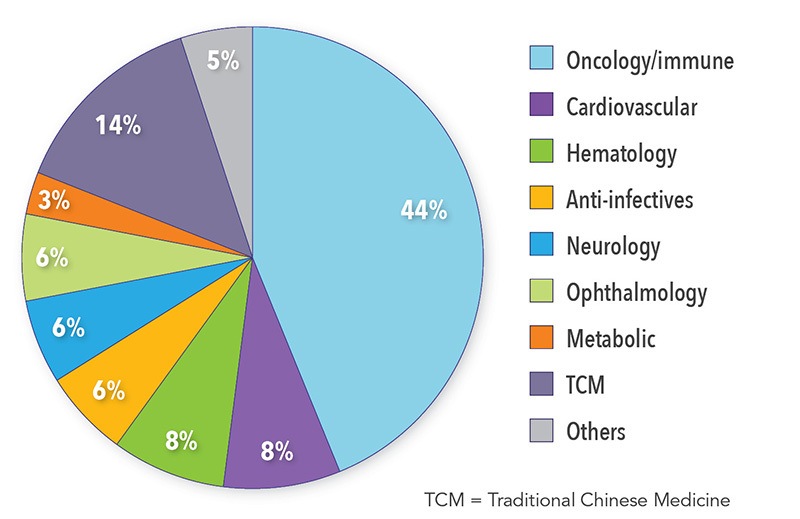
Among the drugs added to the 2017 NRDL, 36 were innovative patent drugs, added after price negotiations with the Ministry of Human Resources and Social Security (MOHRSS) (Note: price negotiation from 2018 onwards has been managed by the National Healthcare Security Administration, NHSA). Of the 36 drugs, 31 were non-TCM (traditional Chinese medicines) drugs including established cancer drugs such as Avastin, Herceptin, Rituxan, and Tarceva.5,6 To negotiate for inclusion, manufacturers were asked to submit an evidence package that included clinical and safety data, prior sales and sales forecasts, and pricing information. For the first time, pharmaceutical companies were able to include health economic evaluation and budget impact analysis as optional submission materials. However, the exact criteria used for assessment were not clearly defined at the policy level and therefore remained a black box for manufacturers in 2017, and the number of pharmacoeconomic experts was too small for sufficient evaluation during the review process.7
The prices of these drugs were cut by an average of 44% compared to their 2016 average retail prices in exchange for being listed in the NRDL. In late 2018, another 17 cancer drugs went through the same process and agreed on discounts to gain national reimbursement. These included 10 medicines approved after 2017.
Regardless of the price cut, the negotiation and NRDL update in 2017 provided an encouraging signal for market access of innovative medicines in China. In general, inclusion on the NRDL is expected to reduce the financial burden on patients and increase access to these innovative therapies. Although the discounted prices translate into reduced profit margins, the increase in the sales volume of these products is expected to offset the reduction in price. The listed prices of Avastin and Herceptin were cut by 61.4% and 64.8%, respectively, after the 2017 NRDL price negotiations; however, the manufacturer still reported that Chinese growth was the main driver in their overall 8% hike in international sales in 2018.8
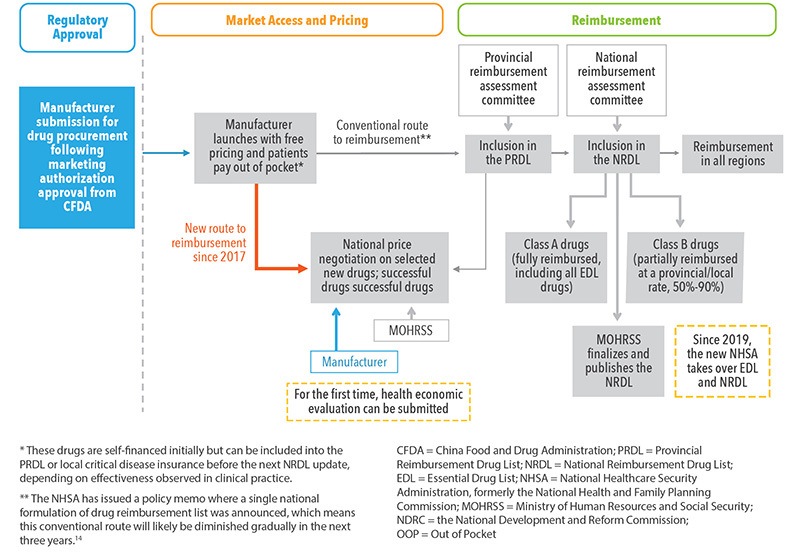
Further details on the types of drugs that are included in the NRDL, as well as the Provincial Reimbursement Drugs List (PRDL), that are reimbursed by public insurance by reimbursable drug categories9 are provided in Figure 2.
The process for market access, pricing, and reimbursement for patent drugs in China, developed as part of the 2017 updates,10 is provided in Figure 3.
Figure 2. Drug Coverage by Public Insurance* (Adapted from MOHRSS3)
2019 NRDL Update: Role of Health Economic Evaluation
The assessment process for the 2019 NRDL update was announced in April 2019. One of the major changes is the adoption of health technology assessment (HTA) before price negotiation for innovative patent drugs. This update also includes further details on the types of economic analysis that would be required to support the submissions.11 Outlined below are the key points in this process.
Eligibility
The drugs eligible for inclusion in the 2019 NRDL needed to have been approved by the National Medical Products Administration (NMPA; previously called China Food and Drug Administration [CFDA]) prior to Dec 31, 2018, with priority consideration given to drugs on the essential drug list (EDL) (i.e., drugs for oncology, ultra-rare diseases, chronic diseases, and pediatric illnesses; and emergency medicines).
Evaluation Process
A key step in the assessment for inclusion is the evaluation of clinical necessity (e.g., unmet need), safety profile, clinical effectiveness, and reasonable pricing for drugs with the same indications, following the pharmacoeconomic principle. Innovative patent drugs, with a much higher price or large potential impact on the health insurance budget, may be included pending price negotiation. In July 2019, it was announced that a revised version of the “China Guidelines for Pharmacoeconomic Evaluation, version 2019” will be published around October 2019. Additionally, a formal HTA agency, the National Center for Evaluation of Medicines and Health Technologies ( 国家药物和卫生技术综合评估中心 ) was established in October 2018 by the National Health Commission (NHC), with branches to be established in the near future focusing on specific disease areas, such as oncology, cardiovascular disease, and pediatrics. An academic center for HTA research will be selected by the NHSA later this year as well.
Expertise
Experts in four main areas are included throughout the entire assessment process.
- Advisory experts (N=300) comment on drug categorization and data analysis, suggest key aspects for technology assessment, and nominate drugs for NRDL inclusion.
- Selection experts (N=25,000) referred from academic and industrial perspectives, including clinicians, pharmacists, and medical insurance management experts from all provinces and all levels of healthcare providers (i.e., from primary care clinics to medical centers). From this expert pool, a certain number are randomly chosen to vote for the drugs for NRDL inclusion (i.e., “selected” drug list). Manufacturers of these selected drugs can then submit the application materials, which should include pharmacoeconomic evaluation (mandatory in 2019 for patent drugs). Note that the manufacturers can only submit application materials for the negotiation stage once their drugs are selected.
- Assessment experts (N=30) provide recommendations based on cost-effectiveness evaluation and budget impact assessment submitted by the manufacturers, only for innovative patent drugs with a high price and a relatively larger impact on budget among the selected drug list.
- Negotiation experts (N=TBD), in conjunction with payers and experts, negotiate directly with manufacturers.
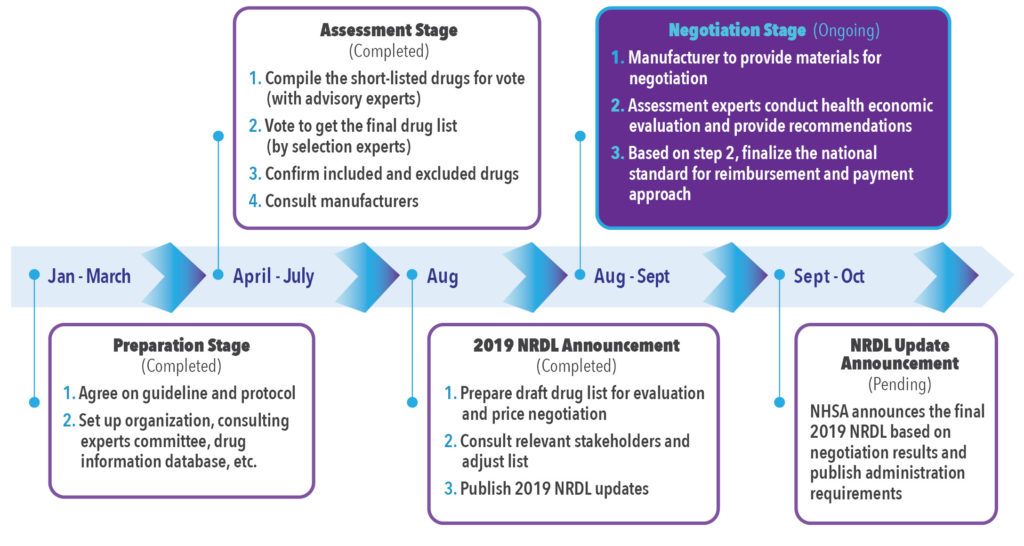
The timeline for the 2019 NRDL assessment process update is shown in Figure 4. The preparation and assessment stages have now been completed and the 2019 updated NRDL was recently published in August 2019.13
- 148 drugs have been newly included without negotiation requirement, which brought the total number of drugs on the NRDL to 2,643, with roughly 50% non-TCM drugs. Meanwhile, several drugs were removed from the list due to their limited value in clinical practice or because better alternatives became available.
- In addition, 128 drugs were selected for price negotiation, of which 109 drugs are patent non-TCM drugs. Along with another 31 patent drugs that were included in the 2017 NRDL and pursuing a deal extension, those selected drugs will enter the negotiation process with the authority.
From 2019 onward, the NRDL will be updated annually with additions and removals of drugs, according to insiders from the authority.14 This is a significant improvement compared to previous updates which took at least four years and rarely removed drugs from the list. This move has seen many manufacturers start preparing for the 2020 NRDL update.
There will likely be a boost in requests for health economic evaluations to be conducted in China, depending on the results of price negotiation and reimbursement evaluation due to be published in October 2019.
Implications, Evidence-Based Strategies, and Key Challenges
Implications
The opportunities for innovative medicines to achieve successful market access and reimbursement have improved and are more predictable than in the past. Established and innovative medicines now have a greater opportunity to quickly reach patients in China. However, the centralized and formalized process means that the NHSA has considerable bargaining power to constrain drug prices and restrict access. As a result, there have been significant price cuts for innovative medicines after price negotiation. The NHSA recently issued a policy memo where a single national formulation of the drug reimbursement list was announced.14 This means the authority to adjust the reimbursement drug list at the provincial level (i.e., PRDL, see Figure 2) may soon be terminated. Provinces will be given a grace period of up to three years before the regulation is fully executed.
Therefore, it is critical for manufacturers of innovative patent drugs to demonstrate the value of their drugs to get reimbursed at the national level moving forward.
Evidence-Based Strategies for Value Demonstration
With a robust evidence package, manufacturers can bring evidence to light that was previously not transparent in decision making in China. The evidence package for value demonstration should include burden of disease analyses, health economic assessment, and pricing models using local data to support the value story.
Proactive Preparation of an Evidence Package
Evidence synthesis and health economic modeling rely on having access to local data, which may take time to collect, so it is vital for pharmaceutical companies to start evidence generation in good time. Important considerations when preparing an evidence package include:
- Developing a strong value proposition and story to successfully communicate the value of treatments. Key components should include disease burden, current treatment and unmet need, clinical value, and economic value.
- Using health economic modeling to generate value evidence package. This would guide value positioning and pricing strategy, connecting local data capacity with health economic tools. These could include, but are not limited to, disease burden simulation to evaluate unmet medical need, cost-effectiveness analysis to demonstrate value to key opinion leaders and payers, and budget impact analysis for the NRDL.
- Leveraging local data resources in real-world evidence (RWE) generation. These studies could focus on disease burden (i.e., including epidemiological study, cost evaluation, and patient research outcomes), treatment patterns, adherence, clinical efficacy, safety, and medical resource utilization. Outcomes from the RWE generation could be used to inform the economic models. Additionally, there are an increasing number of drugs that have been conditionally approved without local clinical trial data, therefore, real-world clinical efficacy and safety data will be required to address the great uncertainties in long-term outcomes.
- Collaborating with academics in support of HTA review through evidence generation projects to facilitate the negotiation process.
Targeted Strategy for Market Access and Pricing
While planning for achieving market access approval, as in many other countries, pricing is a strategic exercise that is best navigated with a thorough understanding of the evidence package, the treatment landscape, and local conditions. Meanwhile, different types of patient access schemes (e.g., charity programs, innovative patient assistant programs, response-based payment) would be introduced to offset the burden of high list prices considering affordability, willingness to pay, and competitors’ pricing.
Initiating evidence generation at the early stage of drug development could help explore and scope an appropriate indication and product profile to target. Following reimbursement, an evidence development plan could also be explored as a risk-sharing method to mitigate the uncertainty of clinical value and risks of uncontrollable budget impact after inclusion.
Key Challenges
This approach is already well established in other countries using HTA, however, there remain challenges associated with this approach in China. These include:
- Availability of local data. Identifying good local data is a challenge as electronic medical records (EMRs), claims databases, and registries are still at an early stage of development and accessibility. Even when data are available from claims and EMRs, they are often not detailed enough to inform economic models. Charts may contain rich data, but collection relies on time-consuming manual extraction. These data challenges hinder effective development of China-specific models or local adaptation of global health economic models.
- Resistance to change. Policy and decision makers are gradually catching up in their knowledge and understanding of health economics and HTA review processes, but since these are methods are still relatively new, there is some resistance to change and a need for continued education.
- Lack of understanding of health economic methodology. There is a lack of HEOR experience among industry and academics. Most have been focused on public health policies and few are expert on quantitative analysis, so the talent pool is insufficient to facilitate the fast-growing demand for HTA review.
These challenges emphasize the importance of robust evidence generation, synthesis, communication, and education.
Conclusions
In summary, given the recent healthcare reforms in China as well as changes in the process for NRDL updates, pharmaceutical manufacturers must tailor their market access strategies and should proactively prepare for evidence synthesis and strategic planning to achieve market access and reimbursement in China. Health economic modeling tools, real-world evidence, and collaboration with local data providers and academics will provide support and facilitate value demonstration and effective pricing negotiation of innovative patent drugs.
We would like to thank Rebecca Zaha, MPH, Senior Director Real-World Evidence China; and, Agnes Benedict, MSc, MA, Executive Director Scientific Affairs, Evidence Synthesis, Modeling & Communication, of Evidera for their insight and expertise in helping to develop and greatly improve the content of this article.
References
- Tan XD, Liu XX, Shao HY. Healthy China 2030: A Vision for Healthcare. Value Health Reg Issues. 2017;12C:112-114.
- Langenbrunner JC, Marquez PV, Wang S. Toward a Healthy and Harmonious Life in China: Stemming the Rising Tide of Non-Communicable Diseases. Human Development Unit; East Asia and Pacific Region. Washington, DC: World Bank. Available at: http://documents.worldbank.org/curated/en/618431468012000892/Toward-a-healthy-and-harmonious-life-in-China-stemming-the-rising-tide-of-non-communicable-diseases. Accessed July 31, 2019.
- Ministry of Human Resources and Social Security of the People’s Republic of China. Notice of the Ministry of Human Resources and Social Security on Printing and Distributing the National Basic Medical Insurance, Work Injury Insurance and Maternity Insurance Drug List (2017 Edition). Available at: http://www.mohrss.gov.cn/yiliaobxs/YILIAOBXSzhengcewenjian/201702/t20170223_266775.html. Accessed July 31, 2019.
- www.gov.cn. The New Medical Insurance Drug List is Published with a Focus on Children’s Medication and Major Disease Medications. Available at: http://www.gov.cn/xinwen/2017-02/23/content_5170341.htm#1. Accessed July 31, 2019.
- People.cn. Drug Access for the First Time “National Negotiations” Medical Insurance Directly to the Pharmaceutical Companies “Bargaining”. Available at: http://finance.people.com.cn/n1/2017/0807/c1004-29453156.html. Accessed July 31, 2019.
- Sina. National Medical Insurance Negotiation Work Completed 8 Varieties Out. Available at: http://med.sina.com/article_detail_103_1_30062.html. Accessed July 31, 2019.
- Koh L, Glaetzer C, Chuen Li S, Zhang M. Health Technology Assessment, International Reference Pricing, and Budget Control Tools from China’s Perspective: What Are the Current Developments and Future Considerations? Value Health Reg Issues. 2016 May;9:15-21. doi: 10.1016/j.vhri.2015.06.004. Epub 2015 Oct 1.
- Reuters. Beset by Copies, Roche Gets Sales Boost from China. Available at: https://www.reuters.com/article/us-roche-results/beset-by-copies-roche-gets-sales-boost-from-china-idUSKCN1MR0GQ. Accessed July 31, 2019.
- Health News. China’s Proposed Medical Insurance Treatment List System. Available at: http://www.xinhuanet.com/health/2019-07/24/c_1124790570.htm. Accessed September 10, 2019.
- Hu J, Mossialos E. China’s Drug Pricing and Compensation Policies and Their Effects. China Health Policy Research. 2017;20(2):5-15.
- National Healthcare Security Administration. Announcement of the National Health Insurance Bureau on the Announcement of the Work Plan for the Adjustment of the National Medical Insurance Drug List in 2019. Available at: http://www.nhsa.gov.cn/art/2019/4/17/art_37_1214.html. Accessed July 31, 2019.
- National Healthcare Security Administration. Notice of the Ministry of Human Resources and Social Security of the State Medical Insurance Bureau on Printing and Distributing the Catalogue of National Basic Medical Insurance, Work Injury Insurance and Maternity Insurance Drugs. Available at: http://www.nhsa.gov.cn/art/2019/8/20/art_37_1666.html. Accessed September 11, 2019.
- Economic Information Daily. Scientifically Constructing a Dynamic Adjustment Mechanism for National Medical Insurance Catalogues – Medical Insurance Catalog Adjustments Can Benefit More Patients. Available at: http://dz.jjckb.cn/www/pages/webpage2009/html/2019-08/09/content_56222.htm. Accessed September 11, 2019.
For more information, please contact us.